HPS: Langdurig statinegebruik
20/12/2011
Deze studie is een follow-up studie van de Heart Protection Study naar effectiviteit en veiligheid van langdurig gebruik van statines. Dit onderzoek richt zich op de langetermijneffectiviteit en -veiligheid van verlaging van het gehalte LDL-cholesterol....
HPS: Effects on 11-year mortality and morbidity of lowering LDL cholesterol with simvastatin for about 5 years in 20,536 high-risk individuals: a randomised controlled trial.Literatuur - HPS Collaborative Group Lancet. 2011 Dec 10;378(9808):2013-20
Heart Protection Study Collaborative Group
Lancet. 2011 Dec 10;378(9808):2013-20.
Achtergrond
Deze studie is een follow-up studie van de Heart Protection Study naar effectiviteit en veiligheid van langdurig gebruik van statines. Dit onderzoek richt zich op de langetermijneffectiviteit en -veiligheid van verlaging van het gehalte LDL-cholesterol met simvastatine 40 mg in de periode tijdens en na het onderzoek. Onderzoek naar de langetermijnveiligheid van statinegebruik is schaars. De mogelijke carcinogene effecten van het realiseren van een (zeer) laag gehalte LDL-cholesterol vormen een belangrijke reden voor zulk veiligheidsonderzoek [1,2].Abstract
BACKGROUND
Findings of large randomised trials have shown that lowering LDL cholesterol with statins reduces vascular morbidity and mortality rapidly, but limited evidence exists about the long-term efficacy and safety of statin treatment. The aim of the extended follow-up of the Heart Protection Study (HPS) is to assess long-term efficacy and safety of lowering LDL cholesterol with statins, and here we report cause-specific mortality and major morbidity in the in-trial and post-trial periods.
METHODS:
20,536 patients at high risk of vascular and non-vascular outcomes were allocated either 40 mg simvastatin daily or placebo, using minimised randomisation. Mean in-trial follow-up was 5•3 years (SD 1•2), and post-trial follow-up of surviving patients yielded a mean total duration of 11•0 years (SD 0•6). The primary outcome of the long-term follow-up of HPS was first post-randomisation major vascular event, and analysis was by intention to treat. This trial is registered with ISRCTN, number 48489393.
FINDINGS:
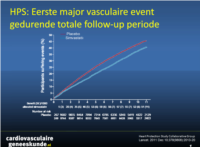
During the in-trial period, allocation to simvastatin yielded an average reduction in LDL cholesterol of 1•0 mmol/L and a proportional decrease in major vascular events of 23% (95% CI 19-28; p<0•0001), with significant divergence each year after the first. During the post-trial period (when statin use and lipid concentrations were similar in both groups), no further significant reductions were noted in either major vascular events (risk ratio [RR] 0•95 [0•89-1•02]) or vascular mortality (0•98 [0•90-1•07]). During the combined in-trial and post-trial periods, no significant differences were recorded in cancer incidence at all sites (0•98 [0•92-1•05]) or any particular site, or in mortality attributed to cancer (1•01 [0•92-1•11]) or to non-vascular causes (0•96 [0•89-1•03]).
INTERPRETATION:
More prolonged LDL-lowering statin treatment produces larger absolute reductions in vascular events. Moreover, even after study treatment stopped in HPS, benefits persisted for at least 5 years without any evidence of emerging hazards. These findings provide further support for the prompt initiation and long-term continuation of statin treatment.
Referenties
1. Davey Smith G, Pekkanen J. Should there be a moratorium on the use of cholesterol lowering drugs? BMJ. 1992;304(6824):431-4.2. Newman TB, Hulley SB. Carcinogenicity of lipid-lowering drugs. JAMA. 1996;275(1):55-60.
3. Kohli P, Cannon CP. Statins and safety: can we finally be reassured? Lancet. 2011;378(9808):1980-1.